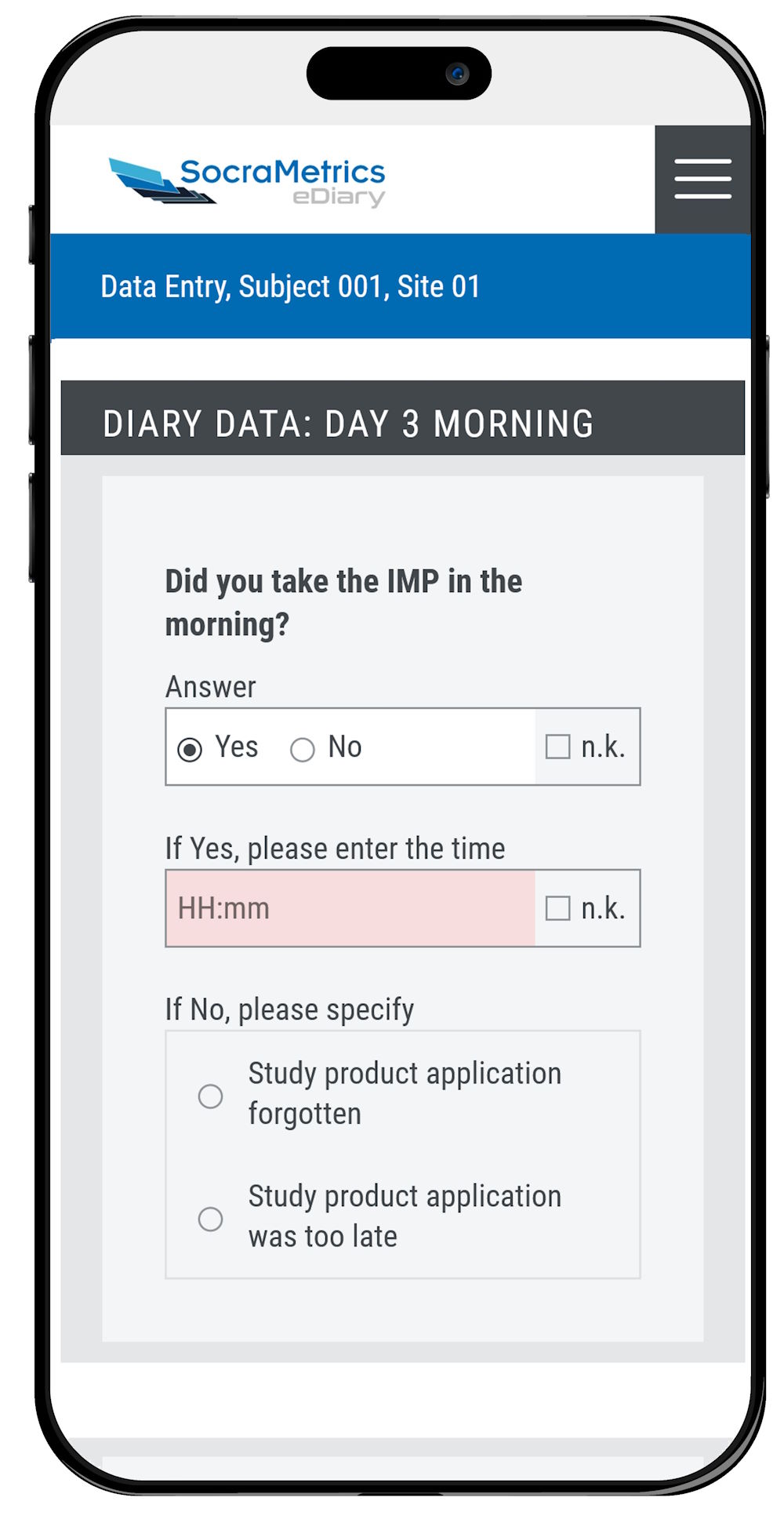
Furthermore, your study participants will appreciate our convenient browser-based eDiary SocraMia!
Volunteers can enter data on their own smartphone, tablet or any other device of their choice.
Data protection and cyber security are both equally important to us. We only collect data that we
really need. This means that participants do not have to disclose any personal information, such as
email addresses to login in to our eDiary SocraMia. We have integrated a sophisticated password
recovery process that eliminates the need to store any non-pseudonymised data.
With SocraMia’s intuitive and clear design, data collection will become part of the daily routine.
Your participants’ compliance will increase!
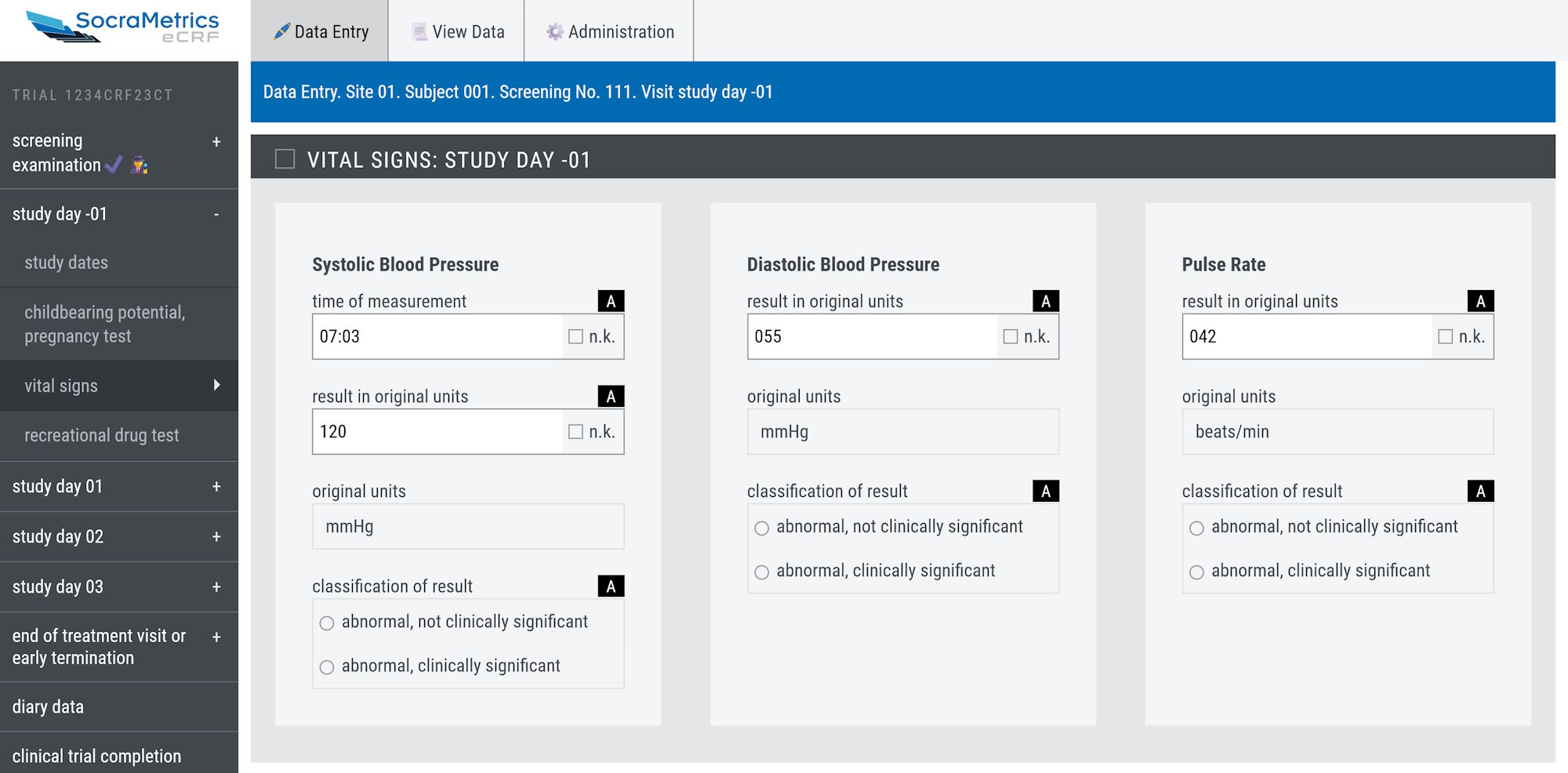
As the eDiary/ePRO is fully integrated into the eCRF system, study staff have real-time access to
all diary data enabling them to perform compliance checks and contact participants promptly in case
of any problems.
And, of course, we can do ePRO, too!